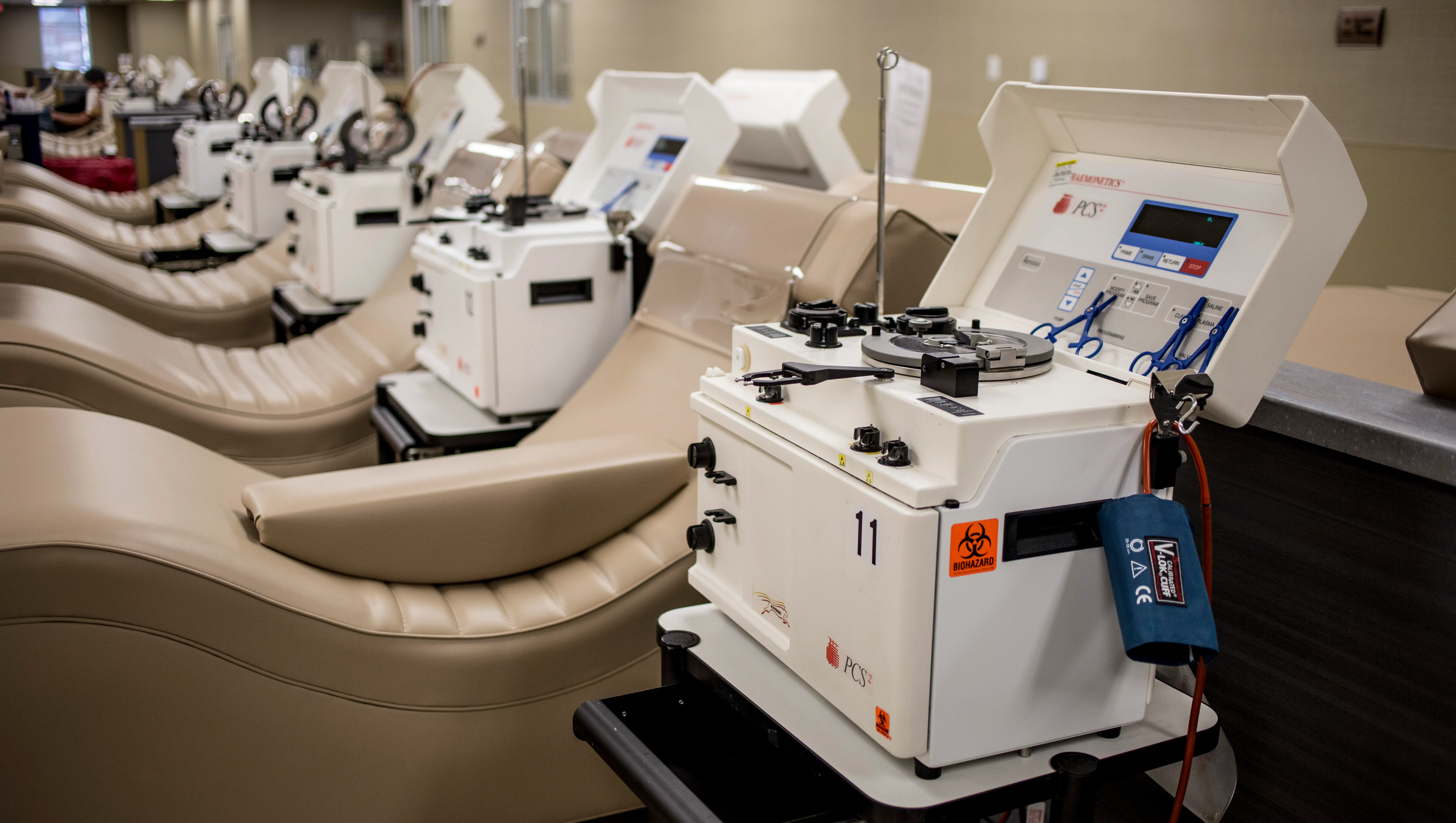
Health Canada has imposed new terms and conditions on all 16 Canadian collection centres operated by plasma company Grifols, after federal inspectors uncovered what they described as "recurring, systemic deficiencies" — a move that comes as the regulator continues to investigate the deaths of two Winnipeg donors.
The two individuals died less than four months apart after donating plasma at separate Grifols locations in Winnipeg. Details surrounding both deaths remain under review by Health Canada.
Head Office Inspection Triggers National Action
The latest regulatory action followed a virtual inspection of Grifols' Canadian head office in Oakville, Ont., which began Jan. 28. While the Oakville facility does not collect plasma directly, it oversees all 16 of the company's Canadian donation sites.
Health Canada said inspectors found a series of troubling issues, including that Grifols was not accurately assessing whether donors were suitable to give plasma, was failing to thoroughly investigate errors and accidents, and did not have a sufficient number of properly trained staff on hand.
Inspectors also found that operating procedures were not consistently followed, and that the company was allowing donations to proceed even when information suggested "the safety of blood could be affected," according to a Health Canada spokesperson.
New Conditions Apply to All Canadian Sites
The terms and conditions imposed by Health Canada apply to all 16 Grifols collection centres across the country. A federal spokesperson said the conditions will remain in effect until the company demonstrates "sustained compliance" with Canadian blood regulations at every licensed location.
Health Canada noted the identified deficiencies are not considered "critical" at this stage, but said it has required "corrective actions" from Grifols and will continue to closely monitor implementation.
In an emailed statement, a Grifols spokesperson said donor safety is the company's "top priority" and confirmed the company is actively addressing the deficiencies identified by Health Canada and putting the new conditions into practice.
Doctor Says System Is Working
At least one prominent Winnipeg physician welcomed the regulatory response. Dr. Ryan Zarychanski, a hematologist at the University of Manitoba who specializes in blood disorders, said the new conditions are a sign that Health Canada's oversight mechanisms are functioning as intended.
"Deficiencies were found, gaps were filled and … [corrective] actions taken. So, I think that's good news for donors in general," Dr. Zarychanski said.
About Grifols
Grifols is a Spanish-based pharmaceutical company that specializes in producing medicines derived from human plasma. The company operates more than a dozen paid plasma collection centres in Canada. Plasma — a protein-rich component of blood — is used in treatments for bleeding disorders, liver disease, and certain cancers, and serves as the raw material for medications such as immunoglobulin.
The investigation into the two Winnipeg deaths remains ongoing. Health Canada has not released timelines for when those reviews are expected to conclude.
Source: CBC Health. This article is based on reporting by Kristin Annable and Michelle Allan of CBC News.